Smart Radiologie – Structured Reporting

Structured Reporting of the Future
medavis demonstrates you in a product study and in the reporting module in medavis RIS the structured reporting of the future. You can then create your reports with predefined structure and controlled terms clearer, more complete and with higher quality. You save time when communication with the referring physician, as the terminology of the report is consistent.
Standardized Format with Structured Entry
When creating structured reports, the reports in the future are not only stored as plain text, but also in the standardized format. This procedure ensures, that all elements of the structured report can be read in a standardized way across providers.
With the structured reporting, medavis RIS builds the bridge between structured document and structured entry. In order to create a structured report, your data is registered already upon entry by selecting terms and codes in a structured way.
Use the advantages of the HTML5 report templates of DRG and RSNA
Standardized requirements by processional committees
Standardized regsitration of examinations including SpeechRecognition and image call
High-quality report templates
Template structure and exchange according to IHE standard
Free text fields for individual issues
Selection and distribution of the templates via repositories on the internet
Selection of elements with speech commands and SpeechRecognition
HTML5 templates offer the possibility to exchange reports in a standardized, provider independent and structured format (IHE-CDA)
HTML5 templates contain elements you can use with a few clicks or speech commands
HTML5 templates can contain logics, that show or hide further elements context-sensitively
More News
medavis Joins Forces with InformMe
medavis Joins Forces with InformMe Hand in Hand for a Digital Workflow in Radiology and BeyondKarlsruhe / Munich, March 25, 2024 – Karlsruhe-based medavis GmbH expands the Group through its acquisition of InformMe GmbH. Taking this step creates the opportunity for the...
The medavis Group Expands its Management Team
Press ReleaseThe medavis Group Expands its Management Team and Sets the Course for Further GrowthThe new and distinctively positioned group, consisting of medavis GmbH headquartered in Karlsruhe, Digithurst GmbH & Co. KG located in Büchenbach, and epiNET GmbH...
medavis users reaffirm their satisfaction with medavis solutions and services
Press Release medavis Users Reaffirm their Satisfaction with medavis Solutions and ServicesWhat are the customers saying? To answer this question, medavis, provider of radiological workflow solutions, once again conducted the periodic customer survey with its users....
Imaging Center, Andernach, Germany: Digitization as the Key to Independent Success
Digitization as the Key to Independent Success Case Study, Imaging Center, Andernach, Germany The economic situation for imaging centers is becoming increasingly challenging despite the steady rise in patient numbers. A lower remuneration, a significant increase in...
New Certificate for Customer Security: booking4med passes pentests with flying colors
Equipped against attacks from the outside booking4med passes pentests with flying colorsFollowing the successful completion of the pentests for the web applications Referrer Portal and Patient Portal, as well as Teleradiology in March, the Appointment Booking Portal...
medavis Teleradiology – Connected Workflows
Teleradiology, integrated reporting from a different site with medavis RIS Connected Working Across Several Sites – Use your full Potential. The medavis RIS to RIS teleradiology solution offers you in the future the possibility to create and send reporting orders...
At the Center: the RIS Report
When Artificial Intelligence meets WorkflowsAt the Center: the RIS Report For radiologists, reporting is the central part of their workflow. Therefore, medavis RIS is consistently optimized for high-performance reporting. It is important that all processes connect...
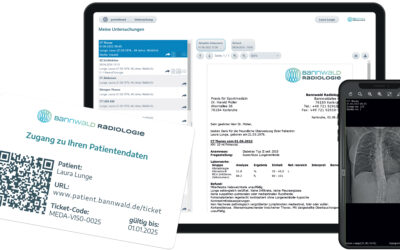
medavis Portal Solutions
medavis Portal Solutions Networking & Communication with Patients and Referrersmedavis Portal Solutions Networking & Communication with Patients and ReferrersIT is of crucial importance for the smooth operation of any radiology department. It is primarily...
medavis Web Applications Pass Pen Tests
medavis Web Applications Pass Pen Tests Security of Patient Datamedavis, leading provider of IT solutions for radiology, has had pen tests conducted on its web applications - Referrer Portal, Patient Portal and Teleradiology. No security-relevant vulnerabilities were...
Data Protection and Data Security
Data Protection and Data Security as a PriorityHigh protection goals such as confidentiality, integrity, and availability of medical data have always had to meet the highest requirements for data security and data protection, even prior to the GDPR coming into...
Continuously Efficient Workflow from a Single Source
Continuously Efficient Workflow from a Single SourceFor many years, the Mühleninsel joint imaging center in Germany has relied on well thought-out, holistic solutions from radiology workflow specialist medavis, which are seamlessly connectedWith the introduction of...
medavis at the IHE Connectathon 2022
medavis as the only RIS providerat the IHE Connectathon IHE profiles XDS.b, XUA and SWF.b successfully tested.From 12 to 16 September, medavis was the only RIS provider represented at the IHE Connectathon in Montreux with four colleagues from Product Management,...
Update of the medavis portal solutions
Update of the medavis portal solutions Deep integration into the radiology workflow with extended functionalities in portal4medmedavis, one of the leading RIS manufacturers in Germany, has completely technically rebuilt its central platform for web applications. More...
medavis presented the new RIS 5 at the German Radiology Congress
Connected into the Future medavis, one of the leading RIS vendors in Germany, successfully presented the new RIS 5 at the German Radiology CongressAt the center of medavis radiology workflow solutions is medavis RIS, which has been one of the most mature and...
New milestones in medavis online appointment booking solution booking4med
New milestones in medavis online appointment booking solution booking4medmedavis, one of the leading RIS manufacturers in Germany, integrates its online appointment booking solution booking4med even deeper into medavis RIS with the latest update. This integration and...
medavis fully supports billing in Switzerland
medavis Fully Supports Billing in SwitzerlandHarmonization and Digitalization of Swiss Payment Traffic The harmonization of Swiss payment traffic to ISO 20022 creates the basis for even simpler and more economical processes. Already since mid-2020, the standardization...
St. Borromeus Hospital, Indonesia: Implementation of Single Data Entry with medavis RIS Improves Workflow Efficiency and Patient Safety
Implementation of Single Data Entry with medavis RIS Improves Workflow Efficiency and Patient Safety Case Study RIS, St. Borromeus Hospital, IndonesiaDigital Radiology Workflow Saint Borromeus Hospital, founded on September 18th in 1921, is one of the oldest hospitals...
Tremendous time savings, Improved work processes, stronger referrer loyalty
Tremendous time savings, Improved work processes, stronger referrer loyalty Case Study booking4med, RADIOLOGY HOHELUFT, Hamburg, GermanyRADIOLOGY HOHELUFT is a modern MVZ with 3 specialists for radiology and nuclear medicine.70% online bookings after a short time...
Appointments Around the Clock
Appointments Around the Clock Case Study booking4med, Radiology Oberland, Hausham, GermanyMore Efficiency in Scheduling Radiology Oberland Improves Processes with booking4med by medavis Nowadays, patients expect short waiting times and high-quality treatment from a...
medavis GmbH and med.essence GmbH form new strategic cooperation
medavis GmbH and med.essence GmbH form new strategic cooperation to develop AI solutionsmedavis will expand its portfolio with interfaces to innovative AI solutions The central future topic of AI is already dominating the digital congresses in radiology. medavis has...
* Mandatory field: Please fill in all fields marked with *.
** Tracking consent: I agree that medavis GmbH can analyze my usage behavior in relation to the newsletter (e.g. which content is clicked on) in order to offer me newsletter content that is tailored to my interests. Further information can be found in our privacy statement. I can revoke this consent at any time with effect for the future by sending an e-mail to marketing@medavis.com or via the link in every e-mail I receive.